 
In brewing beer, calcium chloride is sometimes used to correct mineral deficiencies in the brewing water. Also, it is frequently added to sliced apples to maintain texture. Calcium chloride's freezing-point depression properties are used to slow the freezing of the caramel in caramel-filled chocolate bars. The extremely salty taste of calcium chloride is used to flavor pickles without increasing the food's sodium content. It is commonly used as an electrolyte in sports drinks and other beverages, including bottled water. Īs a firming agent, calcium chloride is used in canned vegetables, in firming soybean curds into tofu and in producing a caviar substitute from vegetable or fruit juices. 
Its use in organic crop production is generally prohibited under the US National Organic Program. It is considered as generally recognized as safe (GRAS) by the U.S. Calcium chloride is permitted as a food additive in the European Union for use as a sequestrant and firming agent with the E number E509. The average intake of calcium chloride as food additives has been estimated to be 160–345 mg/day. Using calcium chloride reduces the need for grading by as much as 50% and the need for fill-in materials as much as 80%. If these are allowed to blow away, the large aggregate begins to shift around and the road breaks down. It keeps the finer dust particles on the road, providing a cushioning layer. A concentrated solution keeps a liquid layer on the surface of dirt roads, which suppresses the formation of dust. The second largest application of calcium chloride exploits its hygroscopic nature and the tackiness of its hydrates calcium chloride is highly hygroscopic and its hydration is an exothermic process. Road surfacing Calcium chloride was sprayed on this road to prevent weathering, giving it a wet appearance even in dry weather. 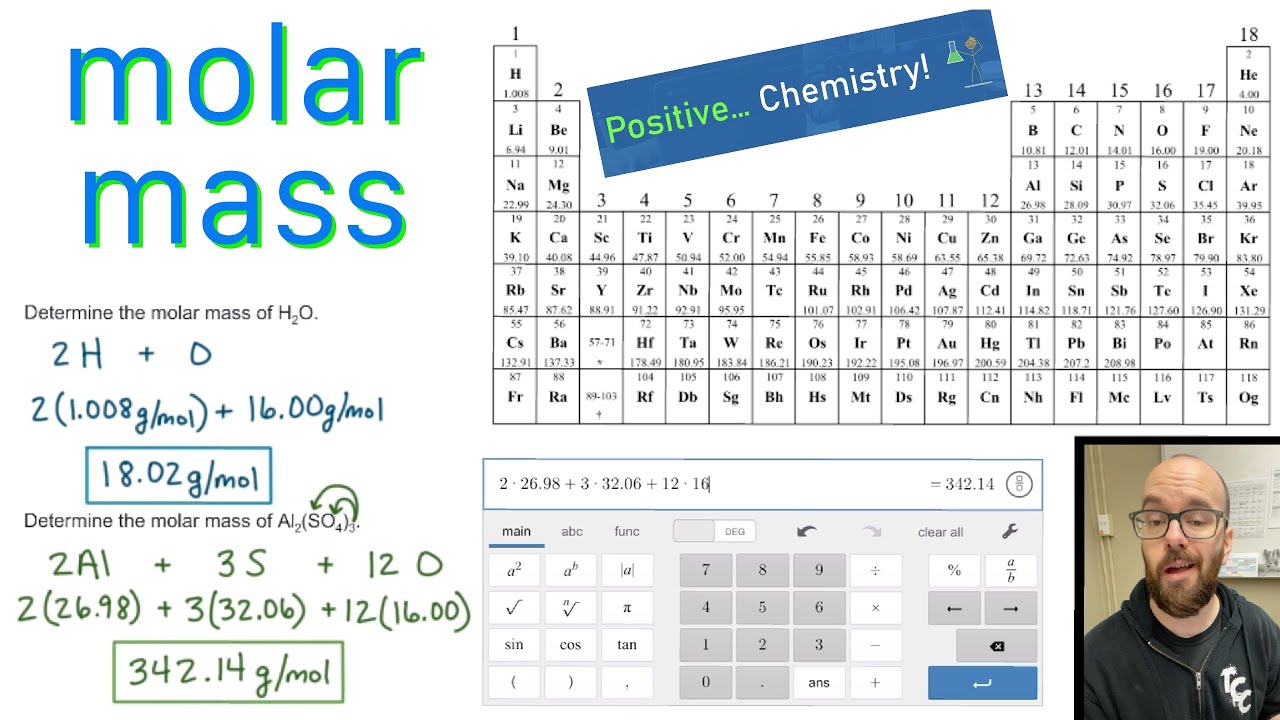
It is also used in domestic and industrial chemical air dehumidifiers. Solutions of calcium chloride can prevent freezing at temperatures as low as −52 ☌ (−62 ☏), making it ideal for filling agricultural implement tires as a liquid ballast, aiding traction in cold climates. When distributed for this use, it usually takes the form of small, white spheres a few millimeters in diameter, called prills. As a deicing agent, it is much more effective at lower temperatures than sodium chloride. Calcium chloride is relatively harmless to plants and soil. This application consumes the greatest amount of calcium chloride. Uses De-icing and freezing-point depression Bulk CaCl 2 for de-icing in Japanīy depressing the freezing point of water, calcium chloride is used to prevent ice formation and is used to de-ice. It was historically called "fixed sal ammoniac" ( Latin: sal ammoniacum fixum ) because it was synthesized during the distillation of ammonium chloride with lime and was nonvolatile (while the former appeared to sublime) in more modern times (18th-19th cc.) it was called "muriate of lime" ( Latin: murias calcis, calcaria muriatica ). Now we interpret the coefficients as referring to molar amounts, not individual molecules.Calcium chloride was apparently discovered in the 15th century but wasn't studied properly until the 18th century. We can leave out the word mol and not write the 1 coefficient (as is our habit), so the final form of the equation, still balanced, is But 6.022 × 10 23 is 1 mol, while 12.044 × 10 23 is 2 mol (and the number is written that way to make this more obvious), so we can simplify this version of the equation by writing it as These coefficients also have the ratio 2:1:2 (check it and see), so this equation is balanced. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
